Efflorescence: 7 Minutes of BS
In this episode of 7 Minutes of BS, we are exploring talk about the salt of the earth and how it can break down masonry structures. We are joined by Sarah Gray, an engineer with RDH Building Science Labs, more earth salt.
What is efflorescence?
efflorescence
ef-luh-res-uh ns | n
The migration of a salt to the surface of a porous material, where it forms a coating.
Origin: French — "to flower out."
“Efflorescence is basically salt that has dried on the surface of brick or stone masonry, and that salt has come from within the masonry itself, and has been pulled to the surface; and that is why you see it as a white deposit that has a salty color and texture.”
The salt is carried by water and deposited on the brick when the water evaporates. The salt comes from the mortar and the brick.
“There are natural water-soluble salts in the mortar, and also brick has some minor amounts of salts from the clays…”
That brick is made from.
“...and then when water gets into a brick wall, those salts can dissolve…”
Into the water.
“...and then as that salty moisture gets pulled to the exterior surface,”
Like when the sun beats on the brick wall,
...the salt is left behind as the moisture evaporates.
Why efflorescence matters
However, if the salt is trapped just beneath the surface of the brick, stone, or block, it can cause further damage because the salt crystals will shrink and swell with wetting and drying cycles.”
And that is called ...
“...subflorescence. And subflorescence can cause the surface of the brick or stone to flake or spall off.”
Wetting, migration, drying, and deposition.
“But if that cycle is not complete, the salts stay within the brick, and those cycles can drive to the point at which crystals become large enough to damage the surface.”
Salt crystals that cannot escape, grow. And they can actually break apart the crystalline structure of the mortar.
When the wetting comes from rain, the efflorescence and subflorescence is manageable. But when it comes from the ground, it gets mighty powerful. Because...
“...the ground will always have some sort of moisture source"
As salt is deposited—and salty water lurks below the surface of the masonry—physics pulls more water into the punchbowl.
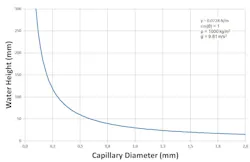
“You definitely get that wicking by either osmosis or capillarity. Pores in masonry are small enough that they have a capillary suction that pulls water from the ground, upward.”
Osmosis is driven by gradients from less salty to more salty, and capillary action is driven by the size of the tube. Capillarity sucks water into the brick, and then osmosis sucks up the salt
Which we are not going to do…
“That’s why the force is so great because that pore size is so small.”
The thinner the space in which the water can travel, the farther up the water can go.
This is one thing high up on a wall getting hit with rain, but it can get ugly where brick is in contact with ground…
“...particularly in a cold climate where we are putting a lot of salts on sidewalks and driveways.”
Reason 541 to make your kid shovel the walk.
All this matters because efflorescence and subflorescence can actually attack the structure of a wall, but also for brick durability
“Durability. A few millimeters of brick sloughing off will not necessarily impact the strength of the wall…”
But,
“...it can impact the durability of the brick itself.”
Because of the structure of the brick
“Brick has a very strong outer layer, called a fire skin, that forms when the clay is fired in the kiln. If that fire skin gets damaged or completely disintegrated by the action of efflorescence and subflorescence, then that brick becomes more exposed.”
Like the soft white underbelly of a brick wall.
“That inner body of clay, although strong in its own right, has a very soft or porous body to it, which can then wick up more water, transport more salt, like an efflorescence snowball effect.”
How to prevent or fix efflorescence:
Mortar is a key part of solving this snowball effect; it is a sort of sacrificial target which actually draws efflorescence away from the snowball.
“Mortar does have a lot of natural salt in it. Calcium silicate, the calcium aluminates that make up the chemical composition of mortar, are typically bound—chemically—and strong. But with excessive amounts of moisture, those bonds can break down, you get dissolution of the salts which can then cause efflorescence.
The other factor is that mortar is typically weaker than the brick around it. Because it’s weaker, it is then more prone to destruction by the salt crystals.”
So one solution to saving brick from efflorescence is to use mortar, specifically mortar that is weaker than the brick, block, or rock.
“...that’s right, it is easier to replace or repair”
Another way to prevent it in the first place is to select brick that has a low salt content
“That can be done at the factory with tests to provide the correct formulation of brick.”
This trick works with mortar, too
“Use a mortar with a low alkalinity and low salt—again, laboratory tests and formulations. And then, as you mix the mortar with water, you want to make sure that it’s clean, potable, or drinkable water. You do not want to use seawater because seawater certainly has salt.”
Aside from choosing the best materials, design can go a long way toward solving problems.
“Detailing the wall assembly of the building such that it is not exposed to a lot of rainwater. That could include detailing ledges, roof caps, flashings, window sills so they do not get water directly resting upon them that can then work through the body of the wall and dissolve the salt.”
Don’t be a dope, slope.
“That’s why flashings are really worth their weight in gold because they help collect, and deflect water away from the wall.”
Regarding the aforementioned de-icing salts and capillary action from the ground, you can use a break between the bottom part of the wall and the rest of the wall.
“A flashing within the wall that serves as a break so that rising damp from capillary action doesn’t get pulled up to higher levels of the wall.”
If you need to retrofit a brick wall, it is difficult to install through flashing at the base. In that case, break out the shovel and
“Dig some soil away from that wall and put in a gravel fill…”
About a foot deep and a foot wide.
“Gravel doesn’t hold moisture very well. If there is no moisture in the soil, it cannot get wicked up into the brick, causing efflorescence.”
A little bit of gravel can make you look prreeettty smart, and that is what we’re here for. Because you get paid for what you do and what you know. And the more you know, the more you can do.
Extra credit:
- Efflorescence - Wikipedia
- Efflorescence: Cause and Control - MasonryInstitute.org
- Efflorescence for Inspectors: International Association of Certified Home Inspectors